top of page

What
Fluxa’s PKM™ software is a web-based application that seamlessly manages product and process specifications throughout the drug development lifecycle. Inherently designed to facilitate collaboration, it enables global sites, functions, CDOs, CMOs, and CDMOs, to work together to scale recipes and execute technology transfers.
Why
Because it is built using modern digital technologies, it provides enterprise-grade security, scalability, and the ability to integrate with other systems such as Enterprise Resource Planning (ERP), Electronic Lab Notebooks (ELN), Laboratory Information Management Systems (LIMS), and Manufacturing Execution Systems (MES).
Due to the company’s confidentiality, the design process, screens, and features have not been displayed in detail

Objectives
Scaling drug development and performing technology transfers from research through commercialization is effort-intensive and error-prone. This is mainly due to the manual way in which disparate processes and data are managed and validated. Current solutions do little to alleviate this problem, leading to silos of knowledge. As a result, significant effort is spent in managing paper-based documentation and associated processes.

Primary Research
Pharma has been undergoing significant change as the industry moves towards more specialty drugs & competitive markets

Increased pressure on Operations
Due to change in Pharma landscape



*Based on annual revenues of $1B - Source: http://www.biopharminternational.com/integrating-technology-transfer-and-facilities-startup-biologics

Problem Statement
Time and operational complexity & knowledge generation increase significantly from Research to Commercialisation

Current solutions do little to alleviate this problem, leading to silos of knowledge

Double Diamond Design Process

Target User Type
The 4W method is an effective approach to define a target user that takes into account 4 Key aspects. Who, What, Where, and When Based on this method we clearly defined our user type.
Secondary
Research

Empathising with the user: Field Study
Understanding the user Type
In order to understand and empathise with the user, I have to understand their needs, goals, and pain points. All that can be done through research, surveys, interviews, and observations. For this research, I used Zoom and even went into the lab to see how they worked.

Observing Behaviours
Understanding the user Type
We conducted interviews with users and discussed with the Analysts who worked with me to identify patterns and problem points.
For reasons of secrecy, I will give the most common example of problems:

Process Control Foundation to accelerate Drug Discovery
This enables the one or more companies to synergize their complimentary solutions to support Life Sciences organizations from design and plan perspective.

Process Control Foundation to accelerate Drug Discovery
This enables the one or more companies to synergize their complimentary solutions to support Life Sciences organizations from design and plan perspective.


Validation
After building the hypothesis, we created paper and interactive prototypes and conducted user testing to validate each of the hypothesis.



Results
After confirming the hypothesis analysis, user testing and multiple design iterations, we were able to move on to the next stage that is building mockup and prototypes
Design Systems - Increasing Scalability
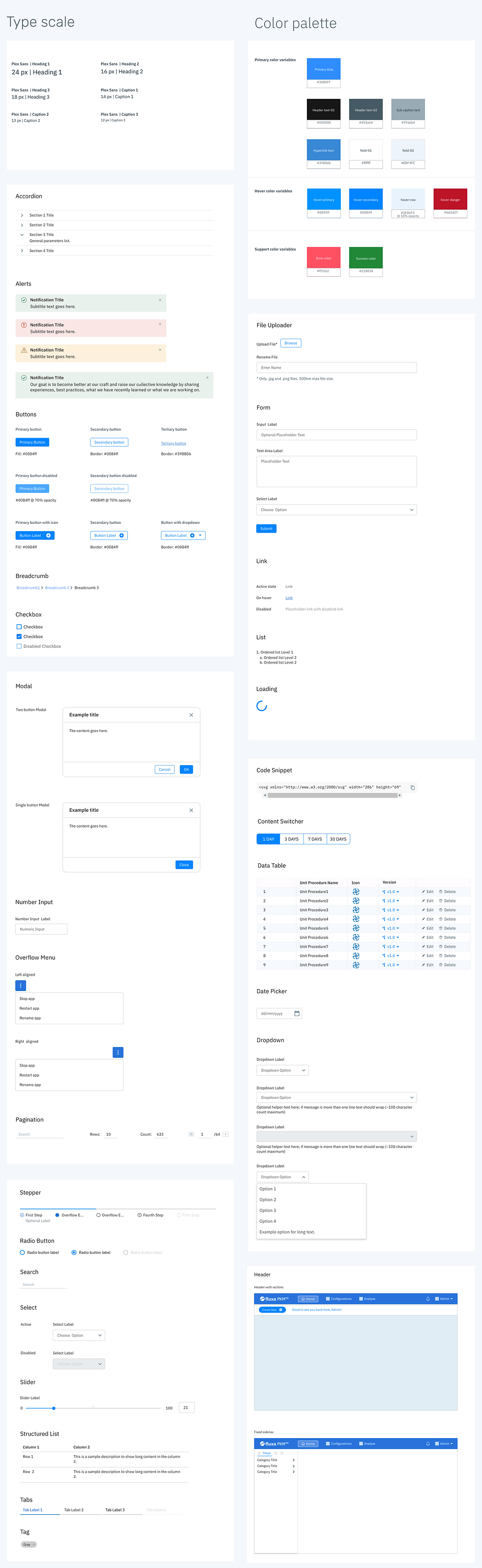

Fluxa PKM Software Features
We articulated an application that seamlessly manages product and process specifications throughout the drug development lifecycle.




Process Outcome - DPMM:L5
Fluxa is working with Biophorum to help biopharmas and CDMOs achieve levels 4 & 5 in the Life Sciences Digital Plant Maturity Model (DPMM), using Fluxa PKM

Connecting Research and
Manufacturing through a single
source of process knowledge
has significant benefits
Connecting Research and Manufacturing through a single source of
process knowledge has significant benefits
Improve collaboration
(e.g. Development <–> Manufacturing )
Faster, cheaper
NPIs & tech transfers
Increase standardization
and compliance
Better understand why
things changed, not just
what changed
Results
After confirming the hypothesis analysis, user testing and multiple design iterations, we were able to move on to the next stage that is building mockup and prototypes



Improved time-to-marke COVID-19 vaccines
Life Sciences Industry Consultant, Alan at Emerson has cited a comparison of a traditional time-to-market versus what was accomplished with the COVID-19 vaccines.


Team Fluxa
PKM became class-defining GMP-validated software that provides a single source of truth for the end-to-end process of scaling and manufacturing a drug. PKM enables pharma companies to create a knowledge continuum from research to manufacturing, closing a critical gap across the industry. It is the world’s first true knowledge management application for drug development and manufacturing that enables ‘one-click’ tech transfers – a game changer for accelerating time to market for new therapies and reducing the cost and effort of transferring drug-making technology within/across biopharmaceutical companies and their external partners like CDMOs and academic or government institutions.


Snaps of team Fluxa at Knowledge Lens Pvt. Ltd Bengaluru
bottom of page
